Nutrition Label Governance.
Nutrition Label Governance

4
1. Introduction
Nutrition label governance refers to the legal and regulatory system that mandates how food businesses disclose nutritional information on packaging. It ensures that consumers receive accurate, clear, and non-misleading information about food products, thereby supporting public health objectives and fair market practices.
It operates through statutes, administrative regulations, and judicial oversight, and is enforced by regulatory authorities such as the Food Safety and Standards Authority of India (FSSAI), the U.S. FDA, and EU institutions.
2. Regulatory Framework
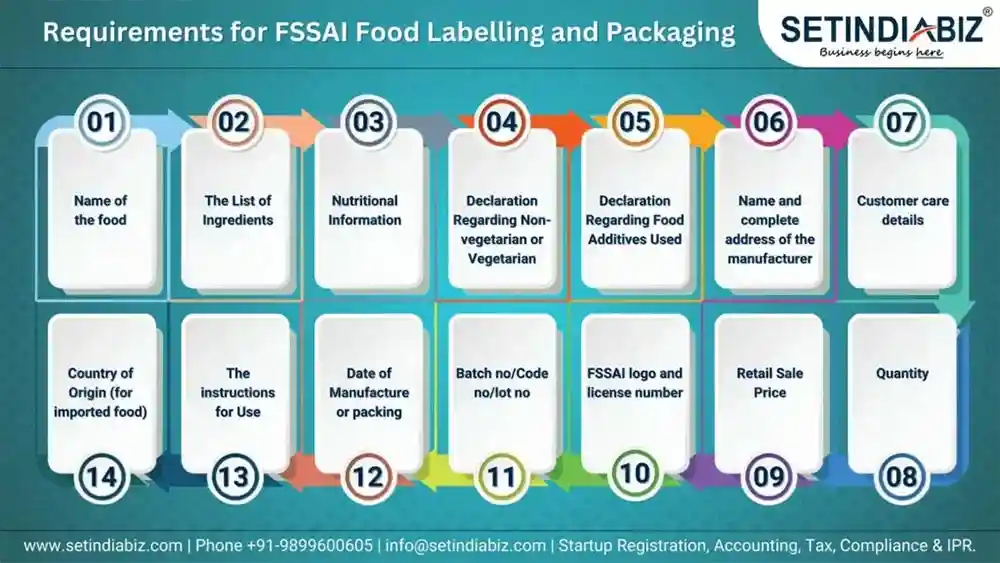
(a) India
- Food Safety and Standards Act, 2006
- Food Safety and Standards (Labelling and Display) Regulations, 2020
Key requirements:
- Nutritional values (energy, fat, sugar, protein, etc.)
- Ingredient list in descending order
- Allergen declarations
- Veg/non-veg symbol
- Manufacturer details and expiry date
(b) United States
- Nutrition Labeling and Education Act (NLEA), 1990
- Standardized Nutrition Facts Panel
- Strict regulation of health and nutrient content claims
(c) European Union
- Regulation (EU) No. 1169/2011
- Mandatory nutrition declaration per 100g/ml
- Clear presentation and legibility requirements
(d) International Standards
- Codex Alimentarius sets global benchmarks
- Influences WTO trade rules and domestic compliance norms
3. Core Principles of Nutrition Label Governance
(i) Transparency
Consumers must be able to clearly understand what they are consuming.
(ii) Accuracy
Nutritional information must be scientifically verified and not exaggerated.
(iii) Non-Deception
Labels must not mislead through false claims, ambiguous wording, or visual presentation.
(iv) Standardization
Uniform labeling formats ensure comparability across products.
(v) Consumer Protection
Recognizes the right to informed choice as a legal and ethical obligation.
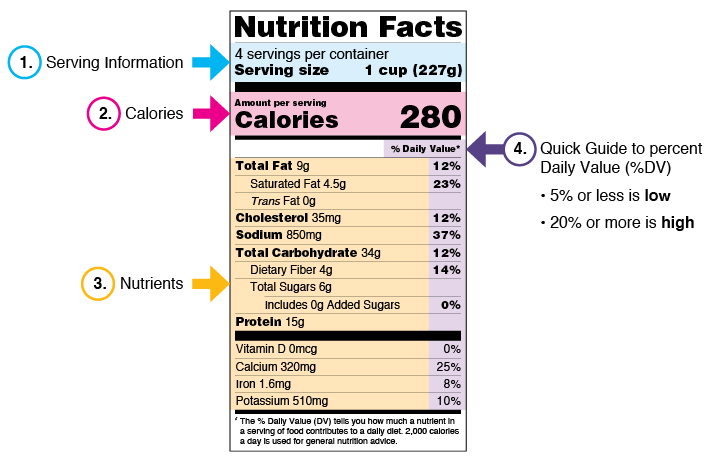
4. Key Components of a Nutrition Label
- Energy (kcal)
- Macronutrients (fat, carbohydrates, protein)
- Micronutrients (vitamins, minerals)
- Sugar and sodium levels
- Serving size
- % Recommended Dietary Allowance (RDA)
- Allergen information
5. Enforcement Mechanisms
- Regulatory audits and inspections
- Product recalls and market withdrawals
- Administrative fines and penalties
- Civil litigation (consumer and competitor suits)
- Criminal prosecution in cases of fraud or harm
6. Major Legal Issues
- Misleading health claims (“natural,” “low-fat,” “sugar-free”)
- Inconsistent global standards
- Online food labeling compliance
- Scientific disputes over nutritional benchmarks
- “Clean label” and marketing-driven ambiguity
7. Important Case Laws
1. Pom Wonderful LLC v. Coca-Cola Co. (2014, US Supreme Court)
- Issue: Misleading labeling of a juice product
- Held: Competitors can sue under the Lanham Act despite FDA regulation
- Significance: Strengthened private enforcement of labeling laws
2. Whitaker v. Thompson (2002, US)
- Issue: Restrictions on health claims
- Held: Qualified health claims permissible with scientific backing
- Significance: Balanced commercial speech and consumer protection
3. National Nutritional Foods Association v. Weinberger (1975, US)
- Issue: FDA’s authority over nutrient labeling
- Held: Upheld regulatory control
- Significance: Confirmed broad administrative powers
4. Teekanne GmbH & Co. KG v. Verbraucherzentrale Bundesverband (2015, CJEU)
- Issue: Misleading fruit tea packaging
- Held: Overall presentation must not mislead consumers
- Significance: Introduced “overall impression test”
5. Nestlé India Ltd. v. Food Safety and Standards Authority of India (2015)
- Issue: Alleged mislabeling and unsafe content (Maggi case)
- Held: Product recall and subsequent compliance review
- Significance: Demonstrated strict enforcement in India
6. PepsiCo India Holdings Pvt. Ltd. v. Food Inspector
- Issue: Misbranding and inaccurate labeling
- Held: Liability for non-compliance upheld
- Significance: Reinforced mandatory disclosure obligations
7. Center for Science in the Public Interest v. FDA
- Issue: Delay in updating nutrition labeling rules
- Held: Courts pressed regulatory updates
- Significance: Ensured regulatory accountability
8. Kraft Foods Group, Inc. Litigation (various consumer suits)
- Issue: Misleading “healthy” and “natural” claims
- Held: Settlements and stricter scrutiny
- Significance: Highlighted consumer protection through litigation
8. Emerging Trends
- Front-of-pack warning labels (high sugar/salt/fat)
- Traffic light labeling systems
- Digital labeling (QR codes)
- Sustainability + nutrition combined disclosures
- AI-based compliance monitoring
9. Conclusion
Nutrition label governance is a cornerstone of food law and public health regulation. It ensures that:
- Consumers are not misled
- Food businesses remain accountable
- Markets function transparently
Judicial decisions across jurisdictions have consistently emphasized truthfulness, clarity, and accountability, making nutrition labeling an increasingly litigation-sensitive and compliance-intensive domain.



comments